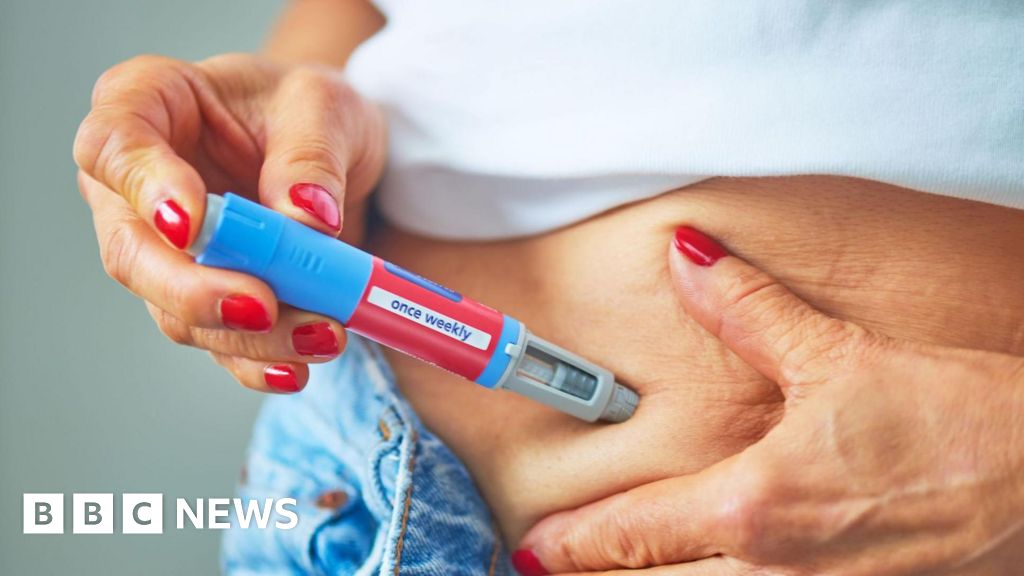
Small risk of severe acute pancreatitis with weight-loss jabs, UK regulator warns
The Guardian • 1/29/2026

Summary
The UK medicines regulator has issued a warning regarding a small risk of severe acute pancreatitis associated with weight-loss medications, specifically GLP-1 drugs like semaglutide and tirzepatide. This advisory follows an increase in reported cases of the condition through the yellow card scheme. Approximately 1.6 million adults in England, Wales, and Scotland utilized these medications for weight management between early 2024 and early 2025. This story highlights the ongoing scrutiny surrounding the safety of popular weight-loss treatments, reflecting broader concerns about the balance between efficacy and potential side effects in pharmaceutical interventions. As obesity rates continue to rise globally, the demand for effective weight-loss solutions grows, making the safety profiles of these drugs critical for public health. Historical parallels can be drawn to past weight-loss products that faced scrutiny after adverse effects were reported, underscoring the importance of vigilant monitoring and transparent communication from health authorities. The implications of this warning extend beyond immediate health concerns, as they may influence patient choices, healthcare provider recommendations, and regulatory practices in the future. As the landscape of obesity treatment evolves, understanding the risks associated with these medications will remain a vital aspect of patient care and public health discourse.
Advertisement
Lindy Score Breakdown (V4.2)
Story Timeline
- 2026-01-29Small risk of severe acute pancreatitis with weight-loss jabs, UK regulator warns (current)
AnalysisRisk 20
Same Story from 3 sources
Breaking Similar stories






Anti-Lindy Similar stories