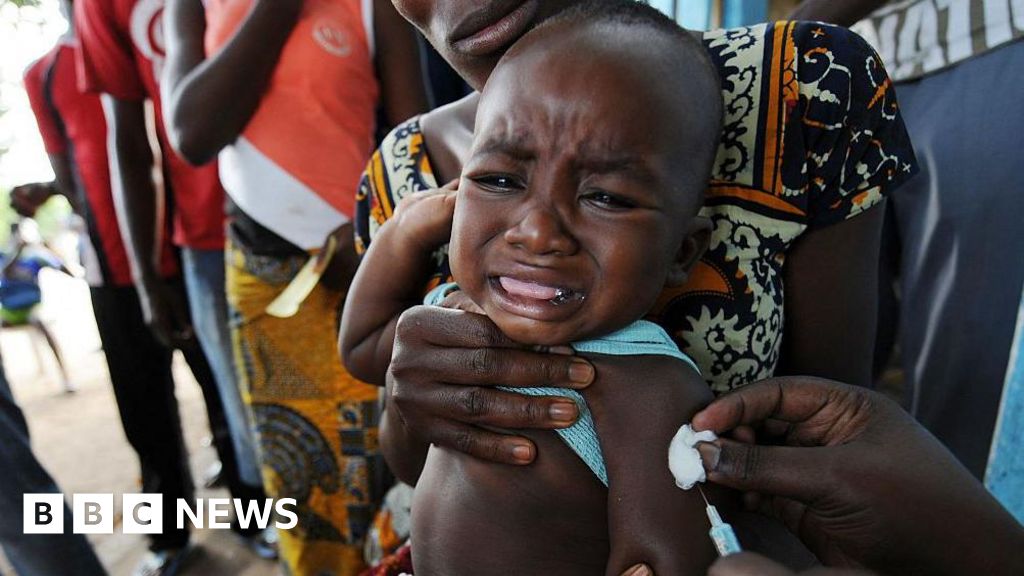
Planned US-funded baby vaccine trial in Guinea-Bissau blasted by WHO
BBC • 2/13/2026 – 2/14/2026
Summary
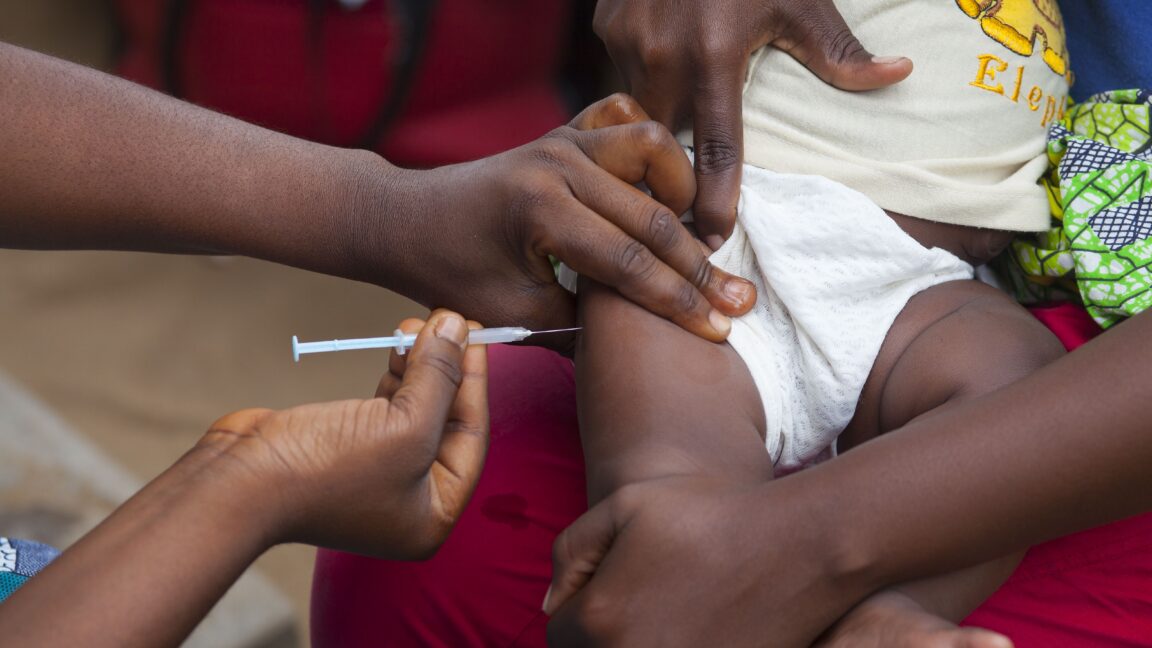
The World Health Organization (WHO) has criticized a planned US-funded vaccine trial in Guinea-Bissau, labeling it as "unethical." The trial involves administering an established hepatitis B treatment to some newborns while withholding it from others. This approach raises significant ethical concerns regarding the equitable treatment of participants in medical research. The study, which has received a $1.6 million research grant from the Centers for Disease Control and Prevention (CDC), aims to examine the effects of a birth dose of the hepatitis B vaccine. However, the WHO's condemnation highlights the ethical implications of conducting a trial that does not provide equal access to a proven vaccine for all newborns involved. The organization emphasizes that such practices are not acceptable in the context of public health research. A Rolling Stone investigation has revealed irregularities in the ethics review process associated with the study. These findings suggest that the approval pathway for the vaccine trial may not have adhered to the stringent ethical standards typically required for research involving vulnerable populations, such as newborns. The controversy surrounding the trial raises questions about the oversight of research practices and the protection of participants in clinical studies. Overall, the situation underscores the complexities and ethical dilemmas faced in conducting medical research in low-resource settings, particularly when it involves life-saving interventions like vaccines. The response from the WHO and the scrutiny from investigative reports indicate a growing concern over the ethical conduct of such trials.
Advertisement
Cluster Activity
Lindy Score Breakdown (V4.2)
Story Timeline
- 2026-02-13
- 2026-02-14Planned US-funded baby vaccine trial in Guinea-Bissau blasted by WHO (current)
- 2026-02-21
AnalysisRisk 20
Same Story from 3 sources
Breaking Similar stories





Anti-Lindy Similar stories